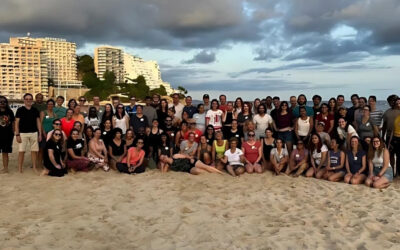P95 exhibits at Bio Asia Pacific 2023
P95 is proud to be an official exhibitor at this year’s Bio Asia Pacific in Bangkok. Bio Asia Pacific is one of the leading conferences and exhibitions for Biotechnology, Life Sciences and Smart Health in Southeast Asia. This year we had the opportunity to present a bit more about our company and our recently created Sout East Asia office in Bangkok by our Regional Director South East Asia, Tharinee Sakhakorn.
Enabling the evaluation of COVID-19 vaccines with correlates of protection
In February 2023, a meeting about correlates of protection (CoPs) against COVID-19 was organized by the International Alliance for Biological Standardization, the European Plotkin Institute for Vaccinology, and Vaccinopolis. The meeting aimed at reviewing the evidence, drawing conclusions, and identifying knowledge gaps.
COVIDRIVE: AZD1222 vaccine effectiveness against hospitalisation
Vaccine effectiveness (VE) studies with long-term follow-up are needed to understand durability of protection against severe COVID-19 outcomes conferred by primary-series vaccination in individuals not receiving boosters. COVIDRIVE is a European public-private partnership evaluating brand-specific vaccine effectiveness (VE). We report a prespecified interim analysis of primary-series AZD1222 (ChAdOx1 nCoV-19) VE.
P95 BV and Ampersand Capital Partners
Leuven, Belgium, May 2nd, 2023 – P95 BV, a global provider of epidemiology and clinical research solutions with a focus on vaccines and infectious diseases, today announced a majority recapitalization by Ampersand Capital Partners, a private equity firm specializing in growth equity investments in the life science and healthcare sectors. Ampersand’s investment will support the growth of P95’s core business and fund the expansion of its CRO services supporting the vaccine and infectious disease end-markets. As part of the transaction, P95 also announced the appointment of Benoit Bouche as Chairman of the company’s Board of Directors.
Lyme Disease Publications
P95 has collaborated in a large review study about Lyme disease. This study has involved databases/surveillance and systematic reviews focusing on Lyme disease incidence, epidemiology, hospitalization, and clinical manifestations, among other outcomes, in different regions of the World.
P95’s fundraising initiatives
As actions speak louder than words, from an internal fundraising initiative, P95 made multiple donations in 2023 that will help charities across the globe.
international infectious disease observatory: A Joint effort from P95 and LOGEX
Healthcare analytics provider LOGEX and P95, a company specialized in epidemiology of infectious diseases, have signed...
Official Opening of P95 Thailand
Almost two years after expanding its operations to Latin America with the creation of P95 Latina, our company has...
P95 annual retreat in Mallorca
At the beginning of October, we held our Annual Retreat in beautiful Mallorca. Around 100 employees were present! This...
Take a tour of our new P95 Latina office
We're excited to share a quick tour of our new P95 Latina office in Bogotá, Colombia, which opened in August 2022. The...
P95 presents 5 posters at the International Conference on Lyme Borreliosis 2022
The P95 Lyme Team (Leah Burn, MPH, Jehidys Montiel Ramos, Aura Victoria Gutiérrez Rabá, Thao Mai Phuong Tran)...
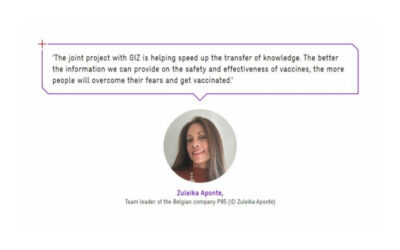
P95 recognized in the GIZ 2021 Integrated Company Report
The Deutsche Gesellschaft für Internationale Zusammenarbeit (GIZ) GmbH's 2021 Integrated Company Report highlights the...
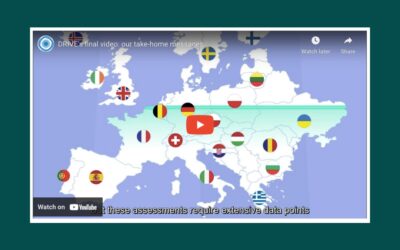
DRIVE project comes to an end: Key Outcomes & Insights
After five years, the Development of Robust and Innovative Effectiveness (DRIVE) project has come to an end. Set up in...
P95 celebrates its 11th birthday
We are celebrating the P95 birthday! On June 16h, 2022, P95 turned 11 years old. To everyone who has allowed our...
P95 Latina team meets in Bogota
On the week of May 16th to May 20th, some of the Epidemiology and Medical Writing team leads met the P95 Latina team...
“We saw in Latin America a hub of talented professionals in epidemiology and research”
Expanding to a different region is the dream of any growing company. For P95, the opportunity arose last year, when...
“I want people to know that P95 has an important role in the improvement of public health worldwide”
Tom Verstraeten, CEO of P95 shares about P95’s first nomination to the Trends Gazelles, recognizing the fastest-growing companies in Belgium
Phase 2b/3 trial HERALD: CVnCoV SARS-CoV-2 mRNA vaccine CANDIDATE
Additional safe and efficacious vaccines are needed to control the COVID-19 pandemic. We aimed to analyse the efficacy and safety of the CVnCoV SARS-CoV-2 mRNA vaccine candidate.
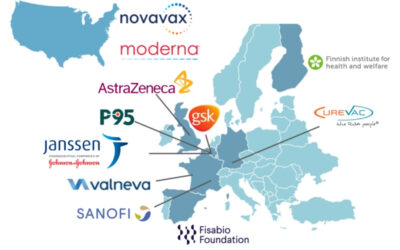
COVIDRIVE welcomes 3 new vaccine companies to the consortium
08/02/2022 – Three new vaccine companies (Moderna, Novavax and Valneva) have joined the COVIDRIVE consortium and officially become COVIDRIVE partners. Moderna, Novavax and Valneva will further contribute to the assessment of the effectiveness of COVID-19 vaccines in Europe to support the region’s public health response and address the vaccine companies’ regulatory obligations.
With the inclusion of these three newcomers, COVIDRIVE, which brings together public institutions, small and medium-sized enterprises and vaccine companies, has a total of 11 partners.
P95 nominated for the Trends Gazelles Flemish Brabant 2022
P95 is one of the nominees for the 2022 edition of Trends Gazelles Flemish Brabant. Trends Gazelles award aims to distinguish the fastest-growing Belgian companies over the past 5 years.